Recent publications are highlighted. For a complete list, see the link to Grae's CV at the end of this page.
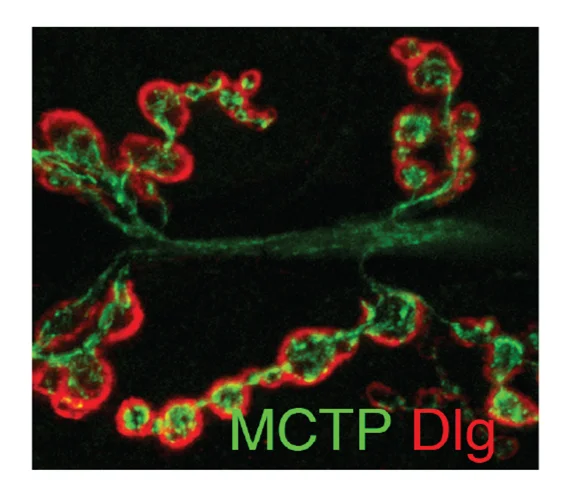
Genç Ö, Dickman DK, Ma W, Tong A, Fetter RD, Davis GW. (2017) MCTP is an ER-resident calcium sensor that stabilizes synaptic transmission and homeostatic plasticity. Elife. May 9: e22904. In a forward genetic screen for mutations that block presynaptic homeostatic plasticity, we identified mctp (Multiple C2 Domain Protein with Two Transmembrane Regions). MCTP localizes to the membranes of the endoplasmic reticulum (ER) that elaborate throughout the soma, dendrites, axon and presynaptic terminal. MCTP is a novel, ER-localized calcium sensor that stabilizes baseline transmission, short-term neurotransmitter release dynamics and homeostatic plasticity.
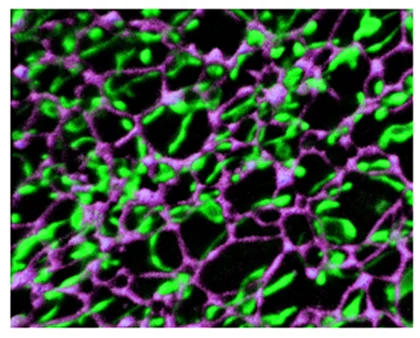
Wang T, Jones RT, Whippen JM, Davis GW. (2016) α2δ-3 Is Required for Rapid Transsynaptic Homeostatic Signaling. Cell Reports 16, 2875-88. The α2δ gene family has been linked to chronic pain, epilepsy, autism, and the action of two psychiatric drugs: gabapentin and pregabalin. Here, we demonstrate that loss of α2δ-3 blocks both the rapid induction and sustained expression of homeostatic plasticity due to a failure to potentiate presynaptic calcium influx and the readily releasable vesicle pool. α2δ proteins reside at the extracellular face of presynaptic release sites throughout the brain, a site ideal for mediating rapid, trans-synaptic homeostatic signaling.
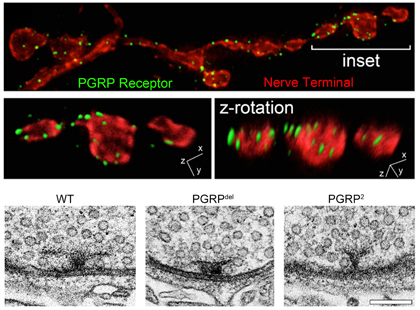
Harris N, Braiser DJ, Dickman DK, Fetter RD, Tong A, Davis GW. (2015) The Innate Immune Receptor PGRP-LC Controls Presynaptic Homeostatic Plasticity. Neuron. 88, 1157-64. The brain is immunologically active. However, it remains generally unknown whether innate immune signaling has a function during the day-to-day regulation of neural function in the absence of pathogens or damage. We identify a novel, neuronal function for an innate immune receptor (PGRP-LC), demonstrating a required during homeostatic synaptic plasticity. PGRP-LC is a candidate receptor for retrograde, trans-synaptic signaling, a novel activity for innate immune signaling in any organism.
Johnson AE, Shu H, Hauswirth AG, Tong A, Davis GW. (2015) VCP-dependent muscle degeneration is linked to defects in a dynamic tubular lysosomal network in vivo. Elife. Jul 13;4. Lysosomes are classically viewed as vesicular structures to which cargos are delivered for degradation. Here, we identify a network of dynamic, tubular lysosomes that extends throughout Drosophila muscle, in vivo. The dynamics and integrity of this tubular lysosomal network requires VCP, an AAA-ATPase that, when mutated, causes degenerative diseases of muscle, bone and neurons. We propose that VCP sustains sarcoplasmic proteostasis by controlling the integrity of a dynamic tubular lysosomal network.
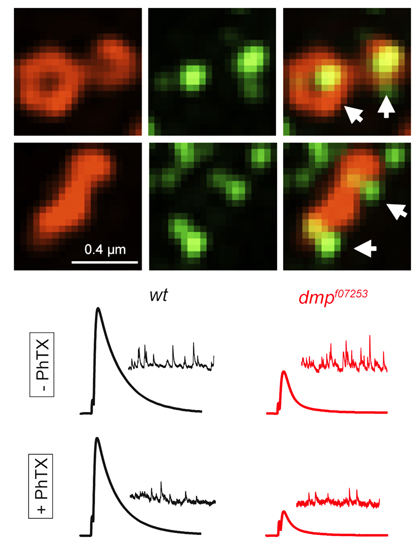
Wang T, Hauswirth AG, Tong A, Dickman DK, Davis GW. (2014) Endostatin is a trans-synaptic signal for homeostatic synaptic plasticity. Neuron 83, 616-29.
Multiplexin is the Drosophila homolog of Collagen XV/XVIII, a matrix protein that can be proteolytically cleaved to release Endostatin, an antiangiogenesis signaling factor, never previously been studied in the nervous system of any organism. We demonstrate that Multiplexin controls calcium channel abundance, presynaptic calcium influx, and neurotransmitter release. Remarkably, release of Endostatin is essential the homeostatic modulation of presynaptic calcium influx and neurotransmitter release. This work identifies a novel form of trans-synaptic signaling during homeostatic plasticity.